The Global Urgency of Stroke Detection
A Growing Worldwide Health Crisis
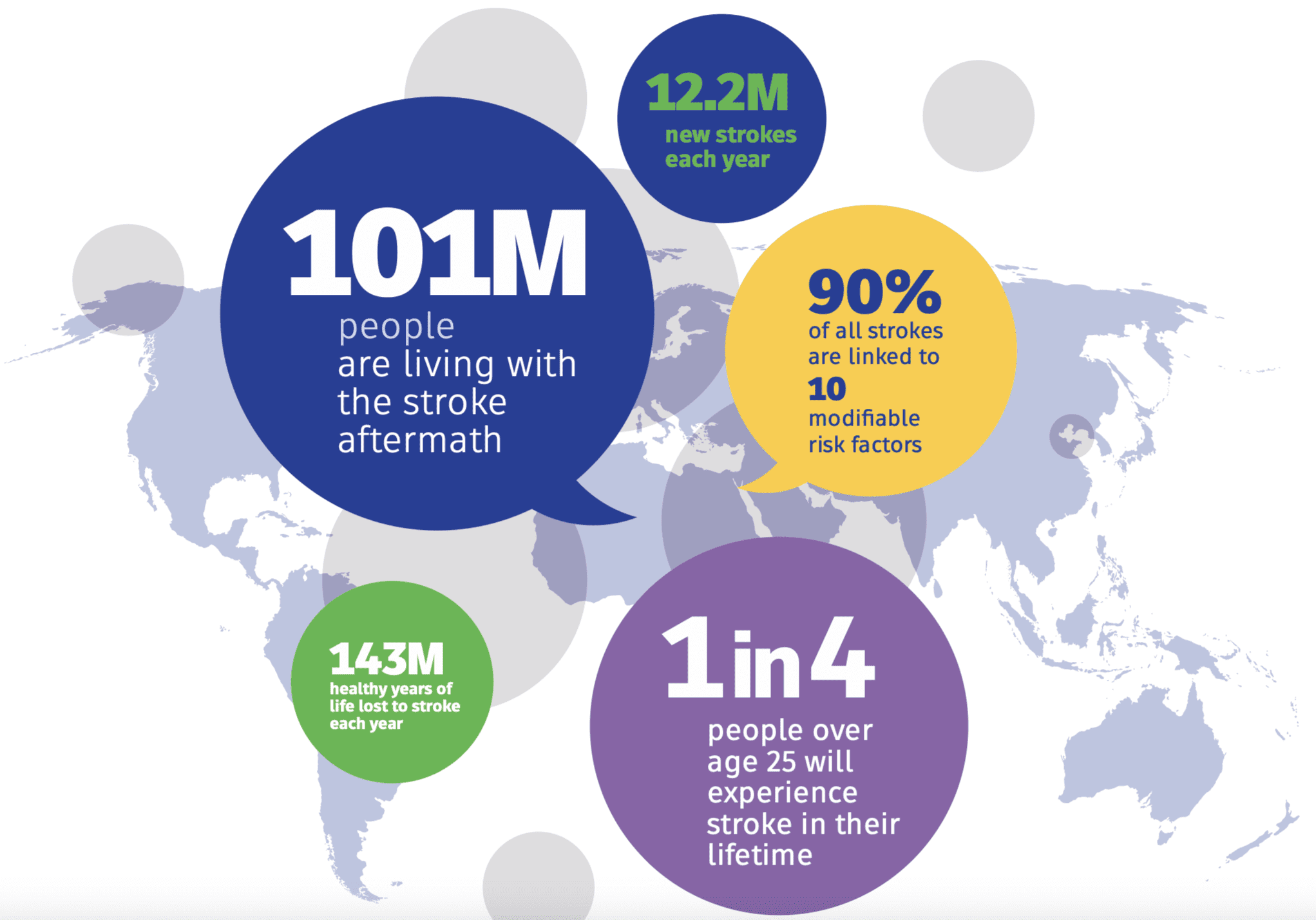
Nearly one in four adults worldwide will experience a stroke in their lifetime. This sobering reality was confirmed by the Global Burden of Disease Study, published in December 2018 in the New England Journal of Medicine. Despite decades of research, the medical community has long awaited a reliable blood test for stroke detection.
Our Primary Mission to Transform Stroke Diagnosis
Why Early Detection Matters
Stroke remains one of the most frequently misdiagnosed medical emergencies. Delays and diagnostic uncertainty cost lives, making accurate and immediate assessment critical.
Our Commitment
Stroke testing is our highest priority. Our mission is to develop, manufacture, and commercialize rapid bedside diagnostic technologies that empower clinicians to make life-saving decisions when every second counts.
Advancing Medical Breakthroughs for Humanity
Galaxy CCRO was founded by clinicians and scientists driven by frustration with the current limitations in stroke management. We are committed to improving outcomes by leveraging under-exploited biomarkers and near-patient diagnostic testing to dramatically increase the speed and accuracy of ischemic stroke diagnosis.
A Mission Rooted in Ethics and Impact
We serve the medical needs of both present and future generations within a morally and ethically responsible environment. We believe our partners must share these values, ensuring that every innovation we bring to market ultimately serves humanity and the greater good.
Intellectual Property Strategy
Galaxy CCRO is dedicated to advancing innovation through a strong advisory ecosystem. We collaborate with respected clinical experts, established industry partners, and a network of experienced associates, supported by a seasoned management team, all operating within a strict ethical research framework.
