Guideline in Action: General Supportive Early Management: Blood Pressure and Glucose
A 68-year-old woman with hypertension, hyperlipidemia, and poorly controlled type 2 diabetes (hemoglobin A1c, 13.8%) presented with acute left arm weakness on waking. Her last known well was the prior night. At an outside hospital, she exhibited left hemiparesis, neglect, and dysarthria (National Institutes of Health Stroke Scale [NIHSS], 7). Noncontrast head computed tomography (CT) showed a right parietal hypodensity, and CT angiography demonstrated a right M2 occlusion (Figure [A] and [B]). She was ineligible for intravenous thrombolysis and was transferred to our facility for endovascular therapy (EVT).
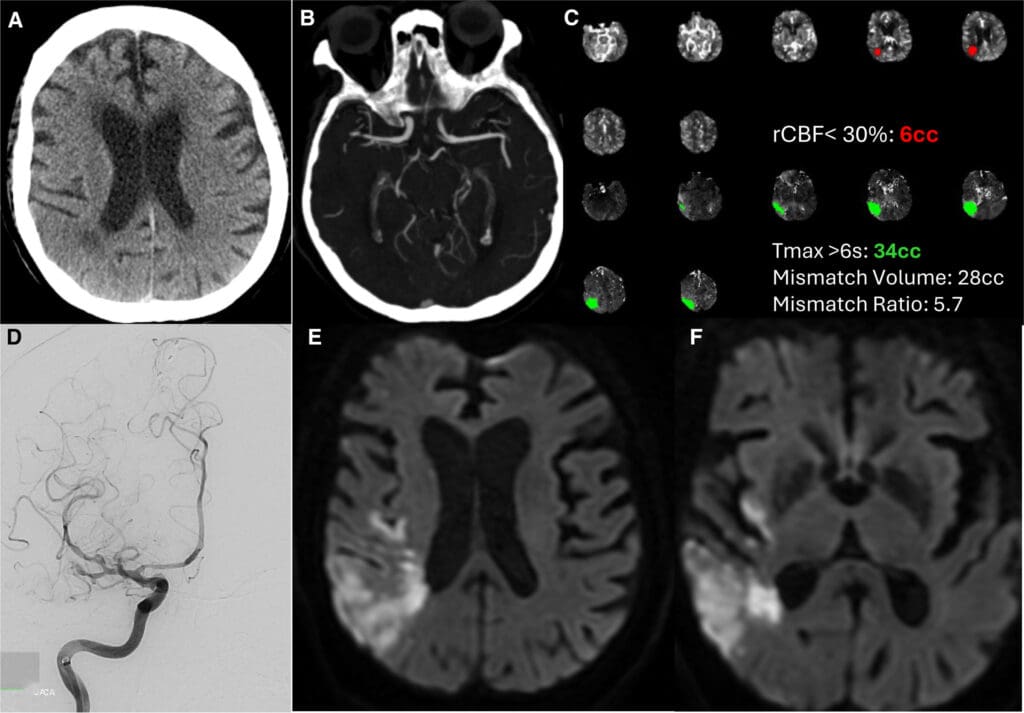
On arrival, her neurological exam worsened (NIHSS, 9) with right gaze deviation, hemianopsia, left facial droop, and persistent left hemiparesis and neglect. Repeat CT confirmed acute right parietal infarction; CT perfusion showed a 34 mL penumbra and a 6 mL infarct core (defined as regional cerebral blood flow <30%; Figure [C]). She underwent EVT with successful reperfusion (Thrombolysis in Cerebral Infarction, 2c), though distal flow remained sluggish (Figure [D]).
Her deficits improved markedly after the procedure (NIHSS, 2). She was severely hyperglycemic (serum glucose, 457 mg/dL) and was started on an insulin infusion, targeting 140 to 180 mg/dL. Approximately 2 hours later, she developed recurrent left-sided weakness and neglect, accompanied by a spontaneous systolic blood pressure drop from 150 to 180 mm Hg to 100 to 110 mm Hg. Electroencephalogram monitoring, initiated during EVT, showed decreased alpha power over the right hemisphere, suggesting hypoperfusion. Norepinephrine infusion was started to maintain systolic blood pressure >150 mm Hg, resulting in rapid improvement (NIHSS, 6→3). By the next morning, she was maintained on low-dose norepinephrine without further deficits. Pressors were weaned off over 24 hours with a stable exam. Magnetic resonance imaging confirmed the right parietal infarction without new lesions (Figure [E] and [F]). She was discharged to acute rehabilitation on day 6 with NIHSS 2 and modified Rankin Scale score of 2.
DISCUSSION
This case highlights the application of the American Heart Association/American Stroke Association Guidelines for the Early Management of Acute Ischemic Stroke for early hemodynamic and metabolic management after thrombectomy.1 The guidelines recommend maintaining BP ≤185/110 mm Hg before the procedure in patients for whom EVT is planned and who have not received intravenous thrombolysis (class 2a, level B-NR).1 The guidelines do not stratify targets by reperfusion grade (class 3: no benefit, level B-R), but it is reasonable to maintain BP ≤180/105 mm Hg during and for 24 hours after EVT (class 2a, level B-NR).1–3 Drug-induced hypertension for perfusion-dependent deficits is not routinely recommended; however, individualized hemodynamic support may be reasonable to correct hypotension or hypovolemia and maintain perfusion in select patients with fluctuating, perfusion-dependent deficits (class 1, level C-EO).1 This patient’s reproducible neurological improvement with systolic blood pressure elevation, supported by neurophysiologic monitoring, underscores the value of dynamic perfusion assessment in real time.
Blood pressure fluctuations are common in acute ischemic stroke, and stabilizing BP while minimizing variability is crucial to preserve penumbral tissue, limit infarct growth, and optimize outcomes. Implementation of standardized clinical pathways improves adherence to evidence-based guidelines and ensures safe, reproducible care. All postreperfusion acute ischemic stroke patients should be managed in ICU or step-down units capable of frequent BP and neurological monitoring.1 Although many patients present with hypertension, acute BP reductions, or excessive variability after reperfusion are associated with larger infarcts and poorer outcomes, underscoring the need to avoid inadvertent hypotension (class 3: no benefit, level B-R).1 In acute ischemic stroke without reperfusion therapy, early intervention to lower BP has not shown clinical benefit and may cause harm, as demonstrated in the CATIS (China Antihypertensive Trial in Acute Ischemic Stroke) and the SCAST (Stroke Controlled Antihypertensive and Stroke Trial),4,5 and reinforced by a meta-analysis.6 These findings support a strategy of permissive hypertension unless BP is severely elevated or another indication exists.2–4 In patients after EVT, the updated guideline specifically discourages intensive BP lowering below 140 mm Hg in the first 24 hours, even in the setting of complete reperfusion, as these thresholds are associated with higher mortality and early neurological deterioration (class 3: no benefit, level B-R).1
The concept of perfusion-dependent neurological deficits dates back decades, from angiographic and xenon-CT studies showing that small systemic BP changes can markedly affect cerebral blood flow in regions with impaired autoregulation.7 Historical clinical series described the use of phenylephrine or dopamine to improve focal deficits, especially in patients with large-vessel occlusion or poor collaterals. Subsequent small trials supported physiological plausibility but not clinical efficacy, and no large, randomized studies have validated outcome benefit.8 Consequently, the guidelines classify that the usefulness of drug-induced hypertension has not been well established and emphasize correction of hypotension and hypovolemia (class 1, level C-EO) to preserve cerebral perfusion.1 Nevertheless, temporary, carefully titrated BP augmentation may be considered in carefully selected cases with clearly demonstrable, reversible perfusion-dependent deficits, supported when possible by neuroimaging or neurophysiologic data (expert opinion). Individualized BP targets should be guided by ongoing neurological assessment of cerebral perfusion.1 Potential risks, including myocardial ischemia, arrhythmia, heart failure exacerbation, arterial dissection, and hemorrhagic transformation of stroke, must be weighed carefully (class 1, level C-EO).1
Take-Home Points
•Avoid hypotension: American Heart Association/American Stroke Association guidelines emphasize correction of hypotension and maintenance of blood pressure ≤180/105 mm Hg after successful reperfusion (class 2a, level B-NR). Acute blood pressure drops and variability are associated with worse outcomes. After EVT, avoid intensive systolic blood pressure targets (<140 mm Hg in the first 24 h) given associations with poorer functional outcomes, higher mortality, and early neurological deterioration (Class 3: Harm, Level A).
•Individualize when appropriate: In select patients with perfusion-dependent deficits, short-term blood pressure augmentation under close monitoring may improve perfusion, though benefits remain unproven.
•Manage glucose carefully: Maintain serum glucose in the 140 to 180 mg/dL range (class 2a, level C-LD); avoid intensive targets (80–130 mg/dL) that increase hypoglycemia risk (class 3: no benefit, level A).
•Build reliable systems: ICU-level monitoring, nurse-driven titration protocols, clear stop rules, and standardized order sets integrated into stroke pathways ensure consistent, safe, and equitable application of care.
Standing order sets that favor short-acting, titratable vasoactive agents, with explicit targets, stop rules, and nursing prompts to prevent overshoot, are essential (expert opinion). Beyond physiology, resource and equity factors also influence real-world implementation. Variability in ICU bed availability, nurse-to-patient ratios, monitoring capacity, and access to subspecialty support can affect adherence to BP and glucose protocols, particularly in resource-limited settings (expert opinion). Scalable, resource-matched order sets with escalation criteria, ongoing education, closed-loop communication, and integration into clinical dashboards, such as Get With The Guidelines, support sustained adherence and quality improvement.
Severe hyperglycemia is also common after ischemic stroke and is associated with worse outcomes.9 Optimal glucose management is likewise clarified in the updated recommendations.1 In this patient, blood glucose was managed with an insulin infusion targeting 140 to 180 mg/dL.1 Although hyperglycemia correlates with hemorrhagic transformation and worse outcomes, intensive glucose control to 80 to 130 mg/dL is not recommended to improve 3-month functional outcomes and prevent severe hypoglycemia risk (class 3: no benefit, level A).10 Instead, treat clearly elevated glucose (eg, ≥180 mg/dL) while avoiding hypoglycemia, using nurse-titrated insulin protocols and frequent checks to maintain safe and consistent control (class 2a, level C-LD).1,10
CONCLUSIONS
This case demonstrates the practical application of American Heart Association/American Stroke Association guidelines for personalized blood pressure and glucose management after reperfusion therapy in acute ischemic stroke. The patient’s reproducible improvement with careful BP augmentation, supported by physiological monitoring, illustrates that individualized, guideline-informed care can be safely implemented when supported by multidisciplinary coordination. Successful execution requires ICU-level monitoring, standardized order sets, and nursing-driven protocols to translate guideline recommendations into patient-centered, outcome-oriented practice.
Acknowledgments
All work was performed at the Divisions of Vascular Neurology and Neurocritical Care, Inova Neuroscience and Spine Institute, Inova Fairfax Medical Campus.
